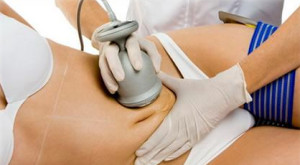
There are millions of dimpled thighs, buttocks and upper arms preventing millions of women from feeling comfortable wearing bathing suits or other revealing garments. Despite following good exercise programs and leading healthy lifestyles, those unattractive pockets of “cottage cheese” remain visible on the surface of the skin.
As a result, non-surgical cellulite removal or body contouring has become increasingly popular in recent years. These procedures do promise to target an endless list of trouble spots without surgery or recovery time … so why not?
The decision is personal, but knowing the difference between procedures may be vital to being pleased with long- and short-term results, among countless other factors. Here’s a look at two very different procedures:
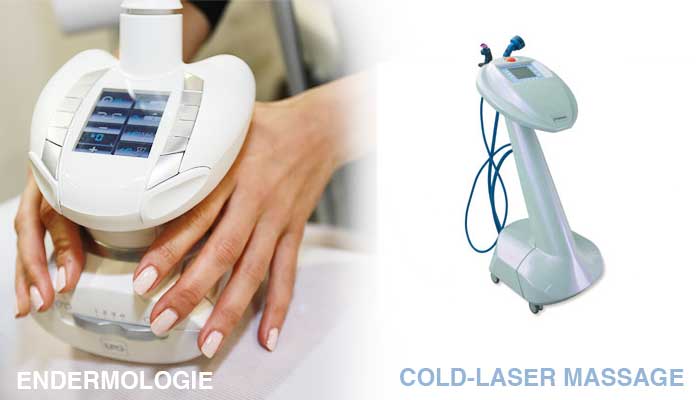
ENDERMOLOGIE BASICS
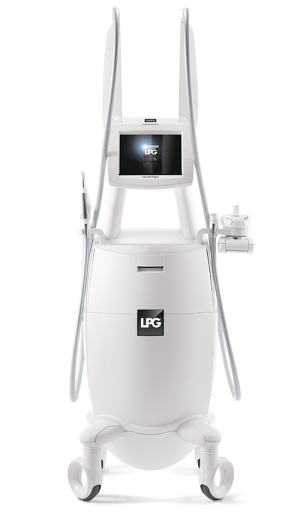
The acknowledged leader in technological developments used in treating the appearance of cellulite, endermologie® was officially born when the first generation Cellu M6 was cleared for use by the U.S. Federal Drug Administration (FDA) in 1988.
Since then, fans have long sworn by the exclusive Mecano Stimulation™ technique that both the skin’s surface. The client lies on a table, while the motorized roll and flap technology suctions and stimulates the skin at varying depths. The speed and direction of the rollers are adjusted depending on the treatment area.
The latest generations of machines feature the Cellu M6 models – both free standing and with a treatment bed. Both products feature the latest technology capable of treating both the body and face.
endermologie® remains high on the list of non-invasive, non-aggressive cellulite treatment options, with little to no adverse effects following treatments.
[button href=”/endermologie-lpg/” colorstart=”#00a24e” colorend=”#00a24e” colortext=”#FFFFFF” icon_size=”12″ class=”” target=”” color=”” align=”vertical” width=”normal” ]Request endermologie® Machine Information[/button]
COLD-LASER MASSAGE BASICS
Like most cellulite treatment machines, the TriActive™ laser was cleared for use by the FDA in 2004 and is also permitted to claim that it “temporarily reduces the appearance of cellulite.” These types of low-level laser devices utilize six low-energy diode lasers and rhythmic suction massage technology to penetrate skin layers.
Non-thermal photons of light penetrate the skin to reach the targeted treatment areas. The light is absorbed by the skin, where it interacts with light sensitive elements within each cell. A cooling agent is used to help maintain the patient’s comfort and reduce the risk of low-level burns.
Cold-laser massages should be avoided by pregnant woman or those suffering from certain medical conditions. Because these procedures involve operating a Class II device (classified by the FDA), they should be administered by a cosmetic physician, spa professional, or aesthetician qualified to administer treatment.